Onyx Motion’s digital coach Swish featured on Canada AM
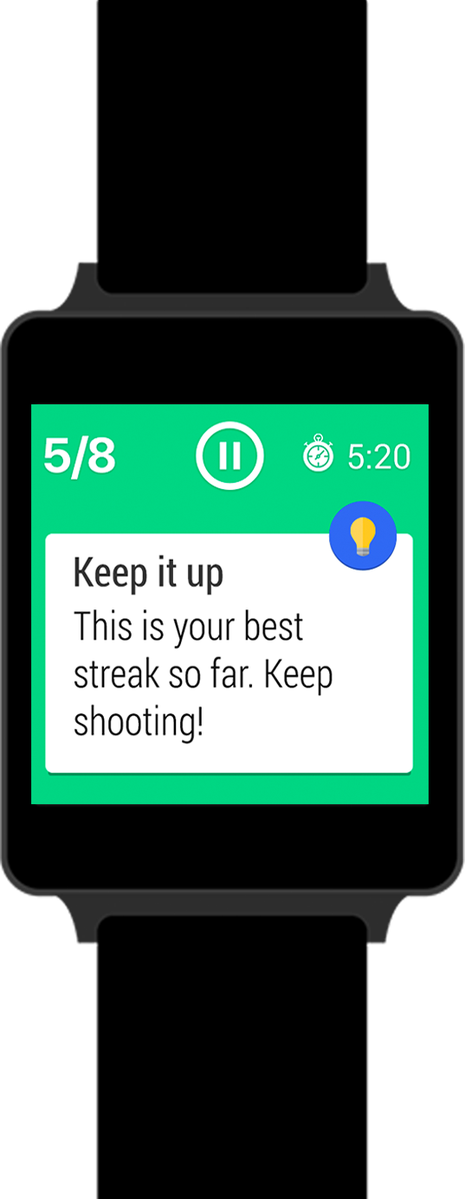
UTEST company Onyx Motion‘s lead product, Swish — a basketball virtual coach that currently works with Android Wear smartwatches to help basketball athletes to improve their shooting technique and emulate their heroes — was featured on Canada AM this morning.
Watch the clip of Onyx Motion’s Swish on Canada AM, CTV’s flagship morning show. Product feature plays after advertisements and begins at 1:40 mark.
Think you’ve got an equally great software-based idea for a start-up? UTEST is currently recruiting for its fourth cohort.
Tom Emrich, a Toronto-based consultant and blogger who specializes in mobile, tablets and wearable technology, presented Swish to Canada AM hosts Marci Ien, Beverly Thomson and Jeff Hutcheson using a Sony SmartWatch 3.
“The application takes your form, measures it and, using the motion sensor of the smartwatch, compares it to experienced players’ data and provides you with a tip,” Emrich explained. “It’s really trying to make sure your next shot is going to be a slam dunk.”
Onyx Motion is part of UTEST’s third cohort. The incubator program, which MaRS Innovation co-directs with the University of Toronto. UTEST is currently recruiting companies to its fourth cohort.





 Nvest
Nvest