LegWorks developing better, more affordable prosthetic knees for developing world
TORONTO (May 27, 2015) – LegWorks Inc., backed with a $2 million blend of private and Government of Canada investments catalyzed by Grand Challenges Canada, is a new Toronto-based company that will contribute to a better life for amputees in developing countries.
The LegWorks AT-knee was covered in the Toronto Star on June 1, 2015 in “Great advances being made in assistive technology” by Kate Allen and in Healio Orthotics & Prosthetics News on June 2.
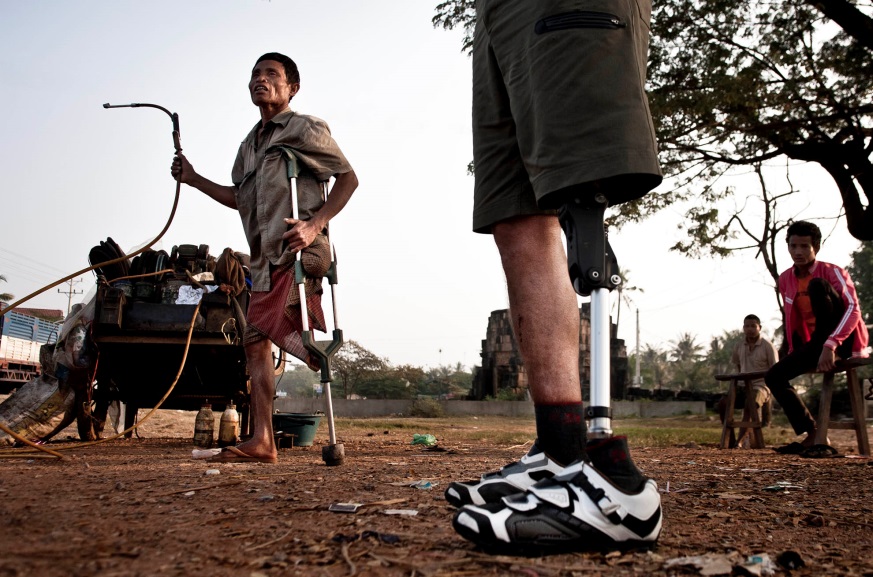
The LegWorks “All-Terrain Knee” (AT-Knee) is a safe, high-functioning, durable, affordable prosthetic knee joint developed at Toronto’s Holland Bloorview Kids Rehabilitation Hospital. It enables lower-limb amputees to walk more efficiently, safely and comfortably. Its patented design provides incredible stability, is easy to fit and maintain, and can even be used in harsh environments, including water.
“With the AT-Knee and LegWorks, it is our goal to begin to provide more universal access to better prosthetic care for individuals living with amputations around the world,” said Jan Andrysek, scientist in the Bloorview Research Institute at Holland Bloorview Kids Rehabilitation Hospital. “We want to make high-quality and well-functioning prosthetic devices affordable and accessible for the many individuals whose needs are currently left unmet.”
In trials, early users in 10 countries reported a 95 per cent preference for the relatively low-cost AT-Knee to more expensive existing technologies. Developed with a $100,000 Grand Challenges Canada seed grant awarded in 2012 to the Bloorview Research Institute, the AT-Knee easily outperformed existing technologies under rigorous conditions in El Salvador, Chile and Myanmar.
 With the new funding, LegWorks will mass produce its innovative, affordable prosthetic knee, the All-Terrain Knee (AT-Knee), the functionality and durability of which makes it ideal for amputees living in the developing world. The $2 million investment deal includes a loan via Grand Challenges Canada of up to $1 million (of which $405,000 has been dispersed), matched by MaRS Innovation, Holland Bloorview Kids Rehabilitation Hospital, Ontario Centres of Excellence and a group of private angel investors. With the $1 million expected from private investors and foundations matched by federal funds, the project will receive an anticipated $2 million to scale-up the success of the company.
With the new funding, LegWorks will mass produce its innovative, affordable prosthetic knee, the All-Terrain Knee (AT-Knee), the functionality and durability of which makes it ideal for amputees living in the developing world. The $2 million investment deal includes a loan via Grand Challenges Canada of up to $1 million (of which $405,000 has been dispersed), matched by MaRS Innovation, Holland Bloorview Kids Rehabilitation Hospital, Ontario Centres of Excellence and a group of private angel investors. With the $1 million expected from private investors and foundations matched by federal funds, the project will receive an anticipated $2 million to scale-up the success of the company.
In the first five years, LegWorks expects 37,000 units to be sold via distributors, NGOs, prosthetic clinics and government rehab facilities, in both high-income countries and the developing world.




